The odour of thiols is notable for being strong and resembling garlic.Īns: Thiophene is a heterocyclic chemical compound. Ques: What is a physical property that distinguishes thiols from other organic compounds?Īns: One of the most basic physical properties that can help a person recognise thiols is their smell. They are similar to alcohol in terms of molecular structure. 
However, instead of an oxygen atom, it possesses a sulphur atom. Thiol is made up of a sulfhydryl group, i.e., Thiol = R-SH.Īns: Thiol is an organic chemical molecule that resembles alcohol and phenols in appearance.Thiol is an organic compound made up of compounds containing a sulphur atom.Thiols are a sulphur analogue of alcohols.Read more : Classification of Organic Compounds Points to Remember They produce cysteine disulphide bridges by oxidising cysteine sulfhydryl groups. Hair curling chemicals, on the other hand, are oxidants. It frees cysteine sulfhydryl groups by reducing cysteine disulphide bridges. Furthermore, the chemical used to straighten hair is a reductant. They are caused by the production of cysteine. Furthermore, disulphide bridges keep such antibody chains, both heavy and light, together.Ĭurls in curly hair are a good example of this. Between various peptide chains, it produces a strong covalent connection. Cysteine must be part of the same peptide chain or subside to the quaternary structure of multi-unit proteins for this to work. Furthermore, oxidation can form cystine units with a disulphide bond when these groups of two cysteine residues in monomers or constituent units are brought close to each other during protein folding (-S-S-).ĭisulphide bonds also have a role in the tertiary structure of proteins. Thiol groups, which are the functional group of the amino acid cysteine, play an important role in biological processes. Sulfonic acids are formed by oxidation with more stronger reagents such as hydrogen peroxide or sodium hypochlorite (RSO3H). Organic disulphide is formed by reagents like bromine (R-S-S-R). This group, as well as the anions it contains, is easily oxidised. With a pKa of 10 to 11, these thiol groups are slightly acidic. Acidity: Thiols have more nucleophilic sulphur atoms than alcohol atoms.Furthermore, thiol groups can form thioether linkages by reacting with vinyl groups. Thioether is formed when a group of thiols reacts with alkenes. Thioacetals, thioethers, and thioesters are all thiols that are comparable to ester, ethers, and acetals. The chemistry of thiols is quite similar to the chemistry of alcohols. As a result, they share some chemical bonding similarities. Sulphur and oxygen belong to the same periodic table group. Reactions: The thiol group of alcohols is a sulphur duplicate of the hydroxyl group (-OH). 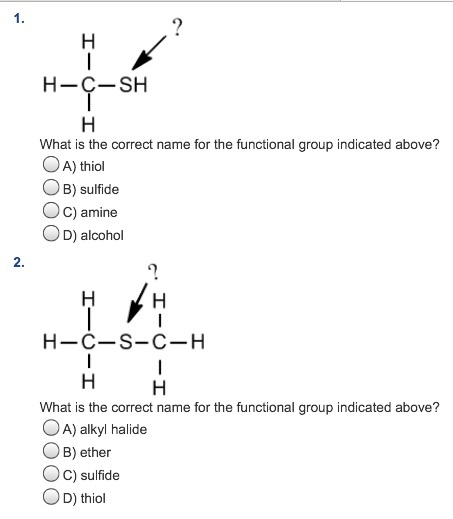
Reducing disulphides with lowering agents like lithium aluminium hydride in dry ether. Heating a halogenoalkane in a sodium hydrosulphide solution:ĬH3CH2Br + NaSH heated in ethanol (aq) → CH3CH2SH + NaBr Synthesis: Thiols are made in the same way that ethers and alcohols are made.Solubility: Thiols are hardly soluble in water.įollowing are the Chemical Properties of Thiol:.Thioether functional groups have similar boiling points and solubility characteristics, however this is not the case with alcohols. Boiling Point: Thiols have lower boiling temperatures than alcohols because they have a small hydrogen bonding connection with water and among themselves.Not all thiols have a foul odour there are numerous thiols that have a pleasant odour. It's also to blame for some wine flaws that have come as a result of unintentional sulphur-yeast reactions. Thiols with a low molecular weight have an unpleasant and pungent odour. Odour: Many thiols have strong odours that resemble the smell of onions and garlic.Following are the Physical Properties of Thiol:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
